
Standard temperature is equal to 0☌ or 32☏ and standard pressure is equal to 1 atm, 101.3kPa or 760 mmHg (torr)ĥ) Electronegativity given by the Pauling scale. Temperature converterģ) Boiling point is the temperature at which a substance changes from liquid to gas state.Ĥ) Density (mass/volume) at standard temperature and pressure, given as gram/cm 3. u = atomic mass unit, 1 u = 1.66 10 -27 kgĢ) Melting point is the temperature at which a substance changes from solid to liquid state.For a more in depth discussion of atomic radius, see the CRC Handbook of Chemistry and Physics, "Atomic Radii of the Elements" and Wikipedia.įurther explanation of the atom may be found here.Elements of the Periodic System - Physical Properties Element Nameġ) Standard average atomic weights (IUPAC 1997) for the isotops naturally present in the element. The Van der Walls radius is used for this periodic table. Two common ones are covalent radius (measures in pirometers) and Van der Waals radius. There are a number of different ways to measure atomic radius.
The Letters, s, p, d, and f designate the shape of the orbital's and the superscript gives the number of electrons in that orbital. This method of notation vastly simplifies the description for large molecules.Įxample: The electron configuration for Be is 1s 22s 2, but we write 2s 2 where is equivalent to all the electron orbital's in the helium atom. Typically, only the outermost electron shells matter in chemistry, so we truncate the inner electron shell notation by replacing the long-hand orbital description with the symbol for a noble gas in brackets. They can predict properties such as stability, boiling point, and conductivity. Using principles of physics, chemists can predict how atoms will react based upon the electron configuration. The electron configuration is the orbital description of the locations of the electrons in an unexcited atom. These atomic weights should be considered provisional since a new isotope with a longer half-life could be produced in the future. Our PTE App is the ultimate tool for every friend of chemistry - whether pupil or teacher, student or professor, amateur or expert, hobbyist or technician.

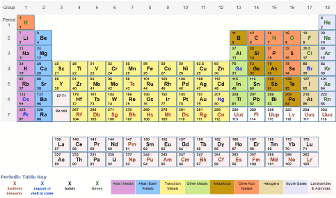
The IUPAC convention is to list the atomic weight of the longest-lived isotope in the periodic table. Immediately find any imaginable information about an element, calculate molar masses of any compounds you like, facilitate your overview with infographics and much more. You may print and distribute as many paper copies of these periodic tables as you want. You will need to use a nice laser printer because the text is quite small. However, for man-made trans-uranium elements there is no "natural" abundance. The following two PDF files can be downloaded and printed on normal 8 1/2 x 11' paper (in landscape orientation) - perfect for including in a 3-ring binder. For naturally-occurring elements, the atomic weight is calculated from averaging the weights of the natural abundances of the isotopes of that element. The average number of neutrons for an element can be found by subtracting the number of protons (atomic number) from the atomic mass.Ītomic weight for elements 93-118. Though individual atoms always have an integer number of atomic mass units, the atomic mass on the periodic table is stated as a decimal number because it is an average of the various isotopes of an element. The standard atomic weight is the average mass of an element in atomic mass units ("amu"). Click here for a list of the elements and their symbols. Typically, a symbol is the truncated name of the element or the truncated Latin name of the element. The molar mass of atoms of an element is given by the relative atomic mass of the element multiplied by the molar mass constant, Mu 1 × 103 kgmol1. In 1869, they independently proposed essentially identical. Weve discussed mass at length by doing molar mass conversions. The atomic symbol is one or two letters chosen to represent an element ("H" for "hydrogen," etc.). The periodic table achieved its modern form through the work of the German chemist Julius Lothar Meyer (18301895) and the Russian chemist Dimitri Mendeleev (18341907), both of whom focused on the relationships between atomic mass and various physical and chemical properties. The number of protons in an atom also determines the chemical behavior of the element. The number of protons in an atom is referred to as the atomic number of that element. For example carbon atoms have six protons, hydrogen atoms have one, and oxygen atoms have eight. The number of protons in an atom defines what element it is. When you open any file of an element in the periodic table, you will find a small table with some basic information about that element. For elements with no stable isotopes, the mass number of the isotope with the longest half-life is in parentheses.
